Based On The Animation, How Many Electron Carriers Are Reduced In The Krebs Cycle Only?
12.four: The Citric Acid Cycle and Electron Transport
- Page ID
- 288909
Learning Objectives
- Depict the reactions of the citric acid cycle.
- Describe the function of the citric acrid wheel and identify the products produced.
- Describe the function of the electron transport chain in energy metabolism.
- Describe the role of oxidative phosphorylation in energy metabolism.
The acetyl grouping enters a cyclic sequence of reactions known collectively every bit the citric acid cycle (or Krebs cycle or tricarboxylic acrid [TCA] bicycle). The cyclical pattern of this complex series of reactions, which bring most the oxidation of the acetyl group of acetyl-CoA to carbon dioxide and h2o, was start proposed by Hans Krebs in 1937. (He was awarded the 1953 Nobel Prize in Physiology or Medicine.) Acetyl-CoA'southward entrance into the citric acid wheel is the beginning of stage Three of catabolism. The citric acid cycle produces adenosine triphosphate (ATP), reduced nicotinamide adenine dinucleotide (NADH), reduced flavin adenine dinucleotide (FADH2), and metabolic intermediates for the synthesis of needed compounds.
Steps of the Citric Acid Bicycle
At first glance, the citric acrid wheel appears rather complex (Figure \(\PageIndex{one}\)). All the reactions, however, are familiar types in organic chemistry: hydration, oxidation, decarboxylation, and hydrolysis. Each reaction of the citric acid cycle is numbered, and in Figure \(\PageIndex{1}\), the two acetyl carbon atoms are highlighted in blood-red. Each intermediate in the bike is a carboxylic acrid, existing as an anion at physiological pH. All the reactions occur inside the mitochondria, which are small-scale organelles within the cells of plants and animals.
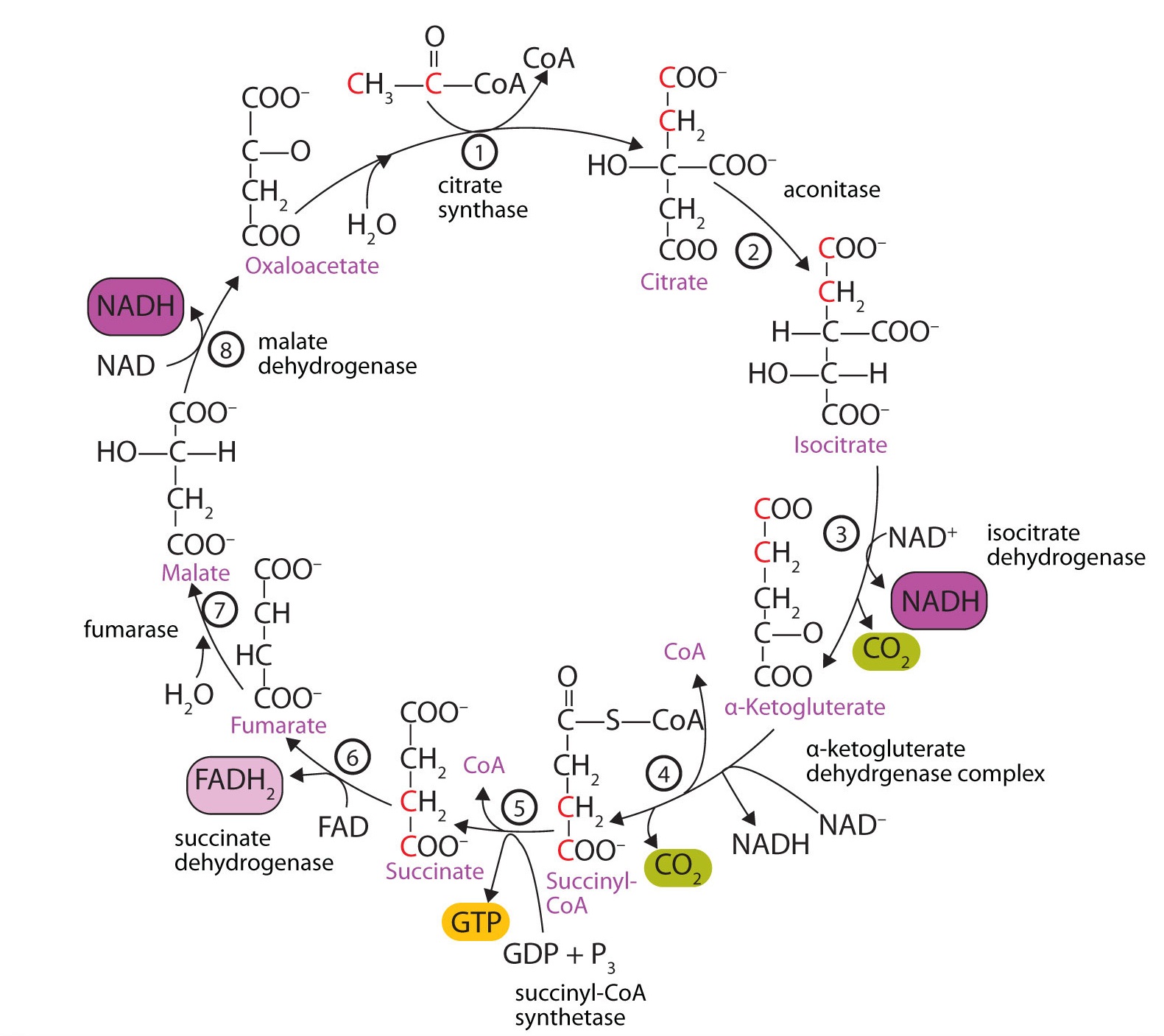
- In the start stride, acetyl-CoA enters the citric acrid cycle, and the acetyl group is transferred onto oxaloacetate, yielding citrate. Note that this footstep releases coenzyme A. The reaction is catalyzed by citrate synthase.
- In the next step, aconitase catalyzes the isomerization of citrate to isocitrate. In this reaction, a tertiary alcohol, which cannot be oxidized, is converted to a secondary alcohol, which can be oxidized in the next pace.
- Isocitrate then undergoes a reaction known as oxidative decarboxylation because the booze is oxidized and the molecule is shortened past one carbon atom with the release of carbon dioxide (decarboxylation). The reaction is catalyzed by isocitrate dehydrogenase, and the product of the reaction is α-ketoglutarate. An important reaction linked to this is the reduction of the coenzyme nicotinamide adenine dinucleotide (NAD+) to NADH. The NADH is ultimately reoxidized, and the energy released is used in the synthesis of ATP, equally we shall see.
- The fourth footstep is another oxidative decarboxylation. This time α-ketoglutarate is converted to succinyl-CoA, and another molecule of NAD+ is reduced to NADH. The α-ketoglutarate dehydrogenase complex catalyzes this reaction. This is the only irreversible reaction in the citric acrid bicycle. Equally such, information technology prevents the bicycle from operating in the reverse direction, in which acetyl-CoA would be synthesized from carbon dioxide.
So far, in the start four steps, two carbon atoms have entered the wheel as an acetyl group, and two carbon atoms have been released as molecules of carbon dioxide. The remaining reactions of the citric acid cycle employ the 4 carbon atoms of the succinyl group to resynthesize a molecule of oxaloacetate, which is the compound needed to combine with an incoming acetyl group and begin another round of the cycle.
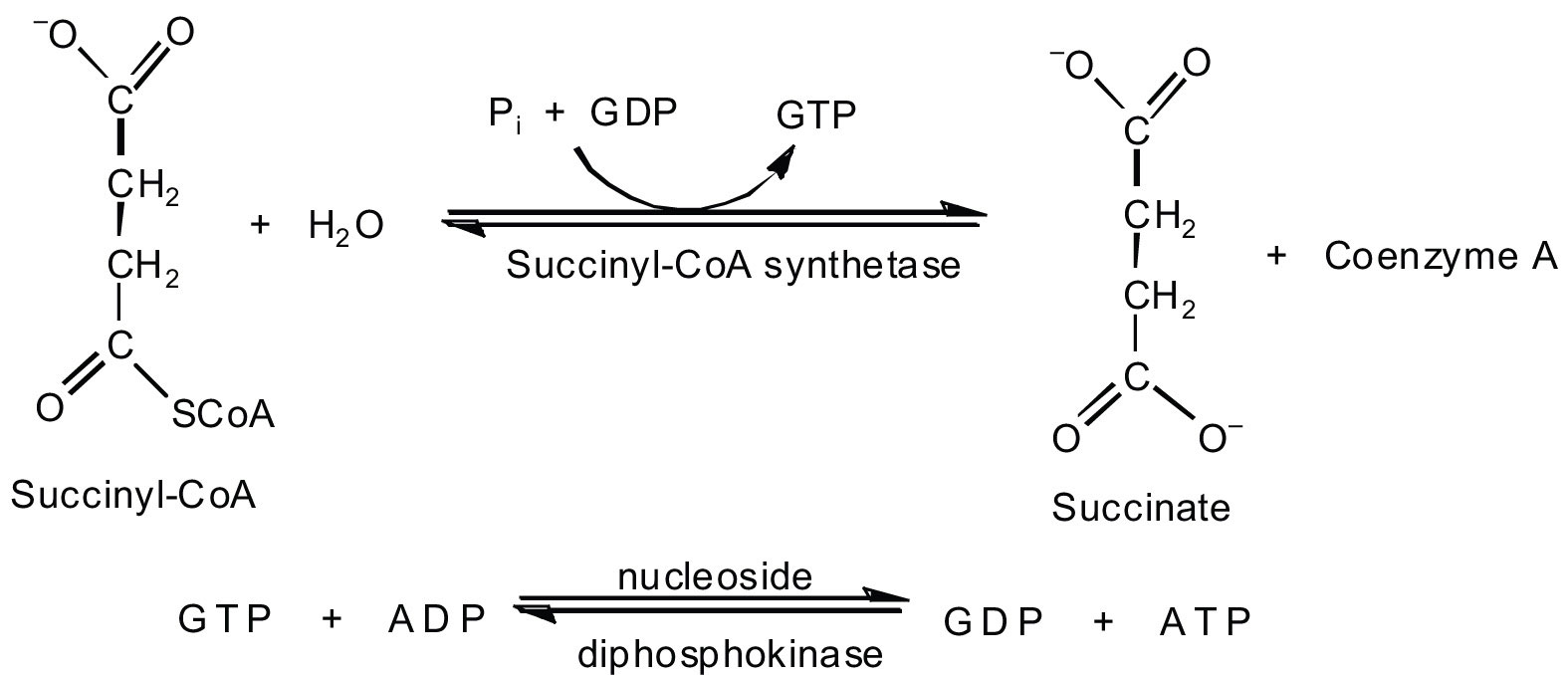
In the fifth reaction, the energy released by the hydrolysis of the high-energy thioester bond of succinyl-CoA is used to class guanosine triphosphate (GTP) from guanosine diphosphate (GDP) and inorganic phosphate in a reaction catalyzed by succinyl-CoA synthetase. This step is the simply reaction in the citric acid bike that straight forms a loftier-energy phosphate chemical compound. GTP can readily transfer its concluding phosphate group to adenosine diphosphate (ADP) to generate ATP in the presence of nucleoside diphosphokinase.
Succinate dehydrogenase then catalyzes the removal of two hydrogen atoms from succinate, forming fumarate. This oxidation-reduction reaction uses flavin adenine dinucleotide (FAD), rather than NAD+, as the oxidizing agent. Succinate dehydrogenase is the only enzyme of the citric acid bike located within the inner mitochondrial membrane. We will see presently the importance of this.
In the post-obit stride, a molecule of water is added to the double bail of fumarate to form L-malate in a reaction catalyzed by fumarase.
One revolution of the cycle is completed with the oxidation of 50-malate to oxaloacetate, brought nearly by malate dehydrogenase. This is the third oxidation-reduction reaction that uses NAD+ equally the oxidizing agent. Oxaloacetate tin can accept an acetyl group from acetyl-CoA, assuasive the cycle to begin again.
Video: "The Citric Acrid Cycle: An Overview". In the matrix of the mitochondrion, the Citric Acid Cycle uses Acetyl CoA molecules to produce energy through 8 chemic reactions. This animation provides an overview of the pathway and its products. NDSU VCell Product's blitheness; for more information delight see http://vcell.ndsu.edu/animations.
Cellular Respiration
Respiration can exist defined every bit the process by which cells oxidize organic molecules in the presence of gaseous oxygen to produce carbon dioxide, water, and energy in the form of ATP. Nosotros have seen that two carbon atoms enter the citric acid wheel from acetyl-CoA (pace ane), and two different carbon atoms leave the cycle every bit carbon dioxide (steps iii and four). Yet nowhere in our discussion of the citric acid cycle have we indicated how oxygen is used. Recall, however, that in the four oxidation-reduction steps occurring in the citric acid bike, the coenzyme NAD+ or FAD is reduced to NADH or FADH2, respectively. Oxygen is needed to reoxidize these coenzymes. Remember, too, that very trivial ATP is obtained straight from the citric acid cycle. Instead, oxygen participation and pregnant ATP production occur subsequent to the citric acrid cycle, in ii pathways that are closely linked: electron transport and oxidative phosphorylation.
All the enzymes and coenzymes for the citric acid cycle, the reoxidation of NADH and FADH2 , and the production of ATP are located in the mitochondria, which are pocket-size, oval organelles with double membranes, oftentimes referred to as the "power plants" of the cell (Figure \(\PageIndex{ii}\)). A prison cell may comprise 100–5,000 mitochondria, depending on its function, and the mitochondria can reproduce themselves if the free energy requirements of the prison cell increase.
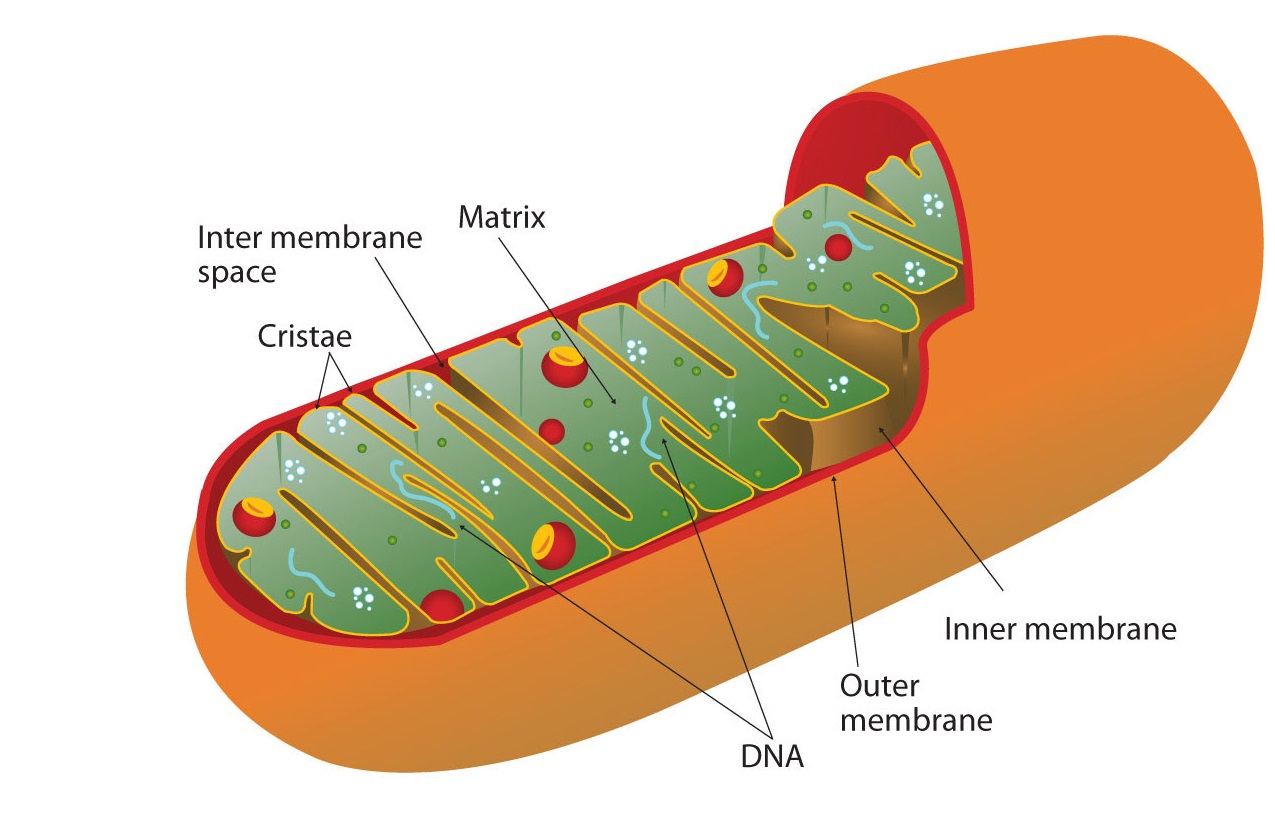
Cellular respiration occurs in the mitochondria
Figure \(\PageIndex{2}\) shows the mitochondrion'due south two membranes: outer and inner. The inner membrane is extensively folded into a serial of internal ridges called cristae. Thus there are two compartments in mitochondria: the intermembrane space, which lies betwixt the membranes, and the matrix, which lies within the inner membrane. The outer membrane is permeable, whereas the inner membrane is impermeable to well-nigh molecules and ions, although water, oxygen, and carbon dioxide tin can freely penetrate both membranes. The matrix contains all the enzymes of the citric acid bicycle with the exception of succinate dehydrogenase, which is embedded in the inner membrane. The enzymes that are needed for the reoxidation of NADH and FADHii and ATP product are also located in the inner membrane. They are arranged in specific positions so that they office in a fashion analogous to a bucket brigade. This highly organized sequence of oxidation-reduction enzymes is known as the electron send chain (or respiratory chain).
Electron Transport
Figure \(\PageIndex{3}\) illustrates the organization of the electron ship chain. The components of the concatenation are organized into iv complexes designated I, Two, Three, and 4. Each circuitous contains several enzymes, other proteins, and metal ions. The metal ions can exist reduced and then oxidized repeatedly as electrons are passed from one component to the next. Recall that a compound is reduced when it gains electrons or hydrogen atoms and is oxidized when information technology loses electrons or hydrogen atoms.
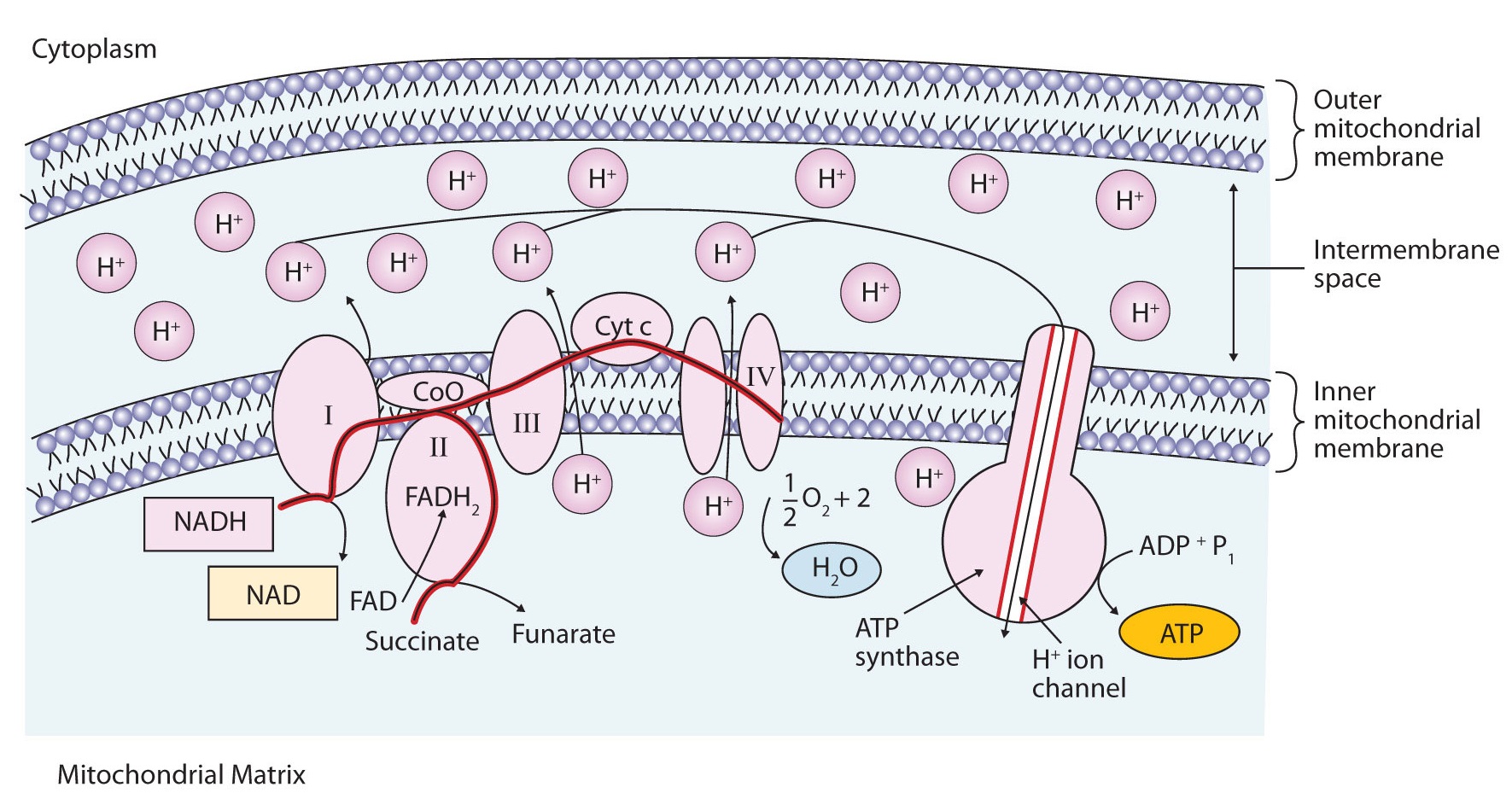
Electrons can enter the electron ship chain through either circuitous I or II. Nosotros will look first at electrons entering at complex I. These electrons come from NADH, which is formed in three reactions of the citric acrid cycle. Let'south apply footstep eight as an example, the reaction in which Fifty-malate is oxidized to oxaloacetate and NAD+ is reduced to NADH. This reaction can be divided into two half reactions:
- Oxidation half-reaction:

- Reduction half-reaction:
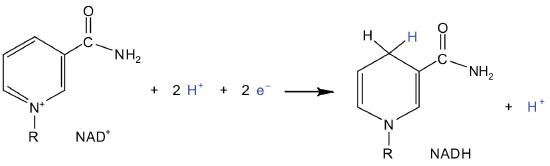
In the oxidation half-reaction, ii hydrogen (H+) ions and 2 electrons are removed from the substrate. In the reduction one-half-reaction, the NAD+ molecule accepts both of those electrons and one of the H+ ions. The other H+ ion is transported from the matrix, across the inner mitochondrial membrane, and into the intermembrane infinite. The NADH diffuses through the matrix and is leap by circuitous I of the electron transport concatenation. In the complex, the coenzyme flavin mononucleotide (FMN) accepts both electrons from NADH. By passing the electrons along, NADH is oxidized back to NAD+ and FMN is reduced to FMNH2 (reduced form of flavin mononucleotide). Again, the reaction can be illustrated by dividing it into its corresponding half-reactions.
- Oxidation half-reaction:
![]()
- Reduction one-half-reaction:
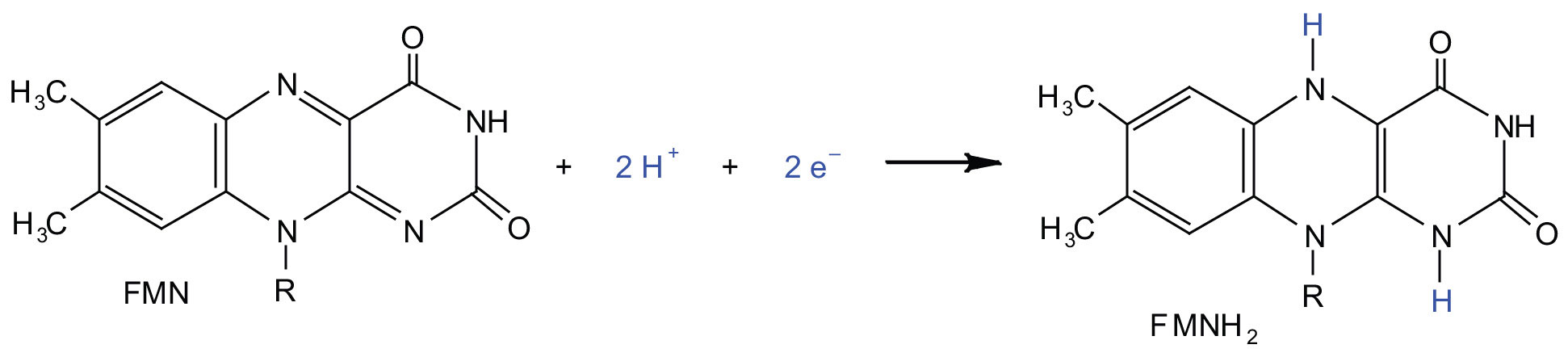
Circuitous I contains several proteins that have atomic number 26-sulfur (Fe·Due south) centers. The electrons that reduced FMN to FMNH2 are at present transferred to these proteins. The iron ions in the Fe·S centers are in the Iron(III) course at first, but by accepting an electron, each ion is reduced to the Fe(II) class. Because each Fe·S center can transfer simply one electron, two centers are needed to take the two electrons that will regenerate FMN.
- Oxidation half-reaction:
\[FMNH_2 → FMN + 2H^+ + 2e^−\]
- Reduction half-reaction:
\[2Fe(Three) \cdot S + 2e^− → 2Fe(II) \cdot Due south\]
Electrons from FADH2, formed in pace half dozen of the citric acrid cycle, enter the electron send concatenation through complex II. Succinate dehydrogenase, the enzyme in the citric acrid bike that catalyzes the germination of FADH2 from FAD is part of circuitous II. The electrons from FADHii are then transferred to an Iron·S protein.
- Oxidation half-reaction:
\[FADH_2 → FAD + 2H^+ + 2e^−\]
Reduction half-reaction:
\[2Fe(III) \cdot S + 2e^− → 2Fe(II) \cdot Due south\]
Electrons from complexes I and Ii are then transferred from the \(Fe \cdot S\) poly peptide to coenzyme Q (CoQ), a mobile electron carrier that acts as the electron shuttle betwixt complexes I or 2 and circuitous III.
Coenzyme Q is also called ubiquinone because it is ubiquitous in living systems.
- Oxidation half-reaction:
\[2Fe(Ii) \cdot S → 2Fe(III) \cdot South + 2e^−\]
- Reduction half-reaction:
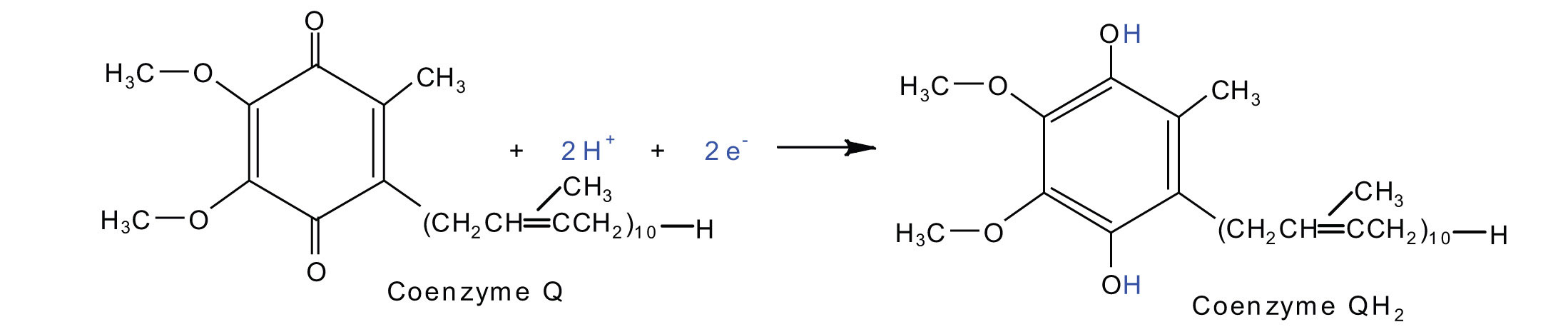
Complexes Three and Four include several fe-containing proteins known equally cytochromes. The iron in these enzymes is located in substructures known as atomic number 26 porphyrins (Effigy \(\PageIndex{4}\)). Like the Fe·S centers, the characteristic characteristic of the cytochromes is the ability of their iron atoms to be as either Atomic number 26(2) or Iron(III). Thus, each cytochrome in its oxidized class—Fe(3)—can accept i electron and be reduced to the Fe(Ii) form. This modify in oxidation country is reversible, so the reduced class can donate its electron to the next cytochrome, and and then on. Complex III contains cytochromes b and c, every bit well as Fe·South proteins, with cytochrome c acting as the electron shuttle betwixt complex III and 4. Complex Iv contains cytochromes a and aiii in an enzyme known as cytochrome oxidase. This enzyme has the power to transfer electrons to molecular oxygen, the terminal electron acceptor in the concatenation of electron transport reactions. In this final step, water (HiiO) is formed.
- Oxidation one-half-reaction:
\[4Cyt\, a_3–Atomic number 26(II) → 4Cyt\, a_3–Fe(3) + 4e^−\]
- Reduction one-half-reaction:
O2 + 4H+ + 4e− → 2H2O

Video: Cellular Respiration (Electron Transport Chain). Cellular respiration occurs in the mitochondria and provides both animals and plants with the energy needed to power other cellular processes. This section covers the electron transport concatenation.NDSU Virtual Jail cell Animations Project animation; ror more than information delight see http://vcell.ndsu.edu/animations
Oxidative Phosphorylation
Each intermediate chemical compound in the electron ship chain is reduced by the addition of one or two electrons in one reaction and then subsequently restored to its original form by delivering the electron(southward) to the adjacent compound along the concatenation. The successive electron transfers result in energy production. But how is this energy used for the synthesis of ATP? The process that links ATP synthesis to the operation of the electron send chain is referred to as oxidative phosphorylation.
Electron transport is tightly coupled to oxidative phosphorylation. The coenzymes NADH and FADH2 are oxidized by the respiratory chain simply if ADP is simultaneously phosphorylated to ATP. The currently accustomed model explaining how these two processes are linked is known equally the chemiosmotic hypothesis, which was proposed by Peter Mitchell, resulting in Mitchell being awarded the 1978 Nobel Prize in Chemistry.
Looking again at Figure \(\PageIndex{3}\), we see that equally electrons are being transferred through the electron transport chain, hydrogen (H+) ions are being transported beyond the inner mitochondrial membrane from the matrix to the intermembrane space. The concentration of H+ is already higher in the intermembrane space than in the matrix, and so energy is required to transport the additional H+ there. This energy comes from the electron transfer reactions in the electron transport concatenation. But how does the extreme difference in H+ concentration so lead to ATP synthesis? The buildup of H+ ions in the intermembrane space results in an H+ ion gradient that is a large energy source, similar h2o behind a dam (because, given the opportunity, the protons will flow out of the intermembrane infinite and into the less concentrated matrix). Electric current inquiry indicates that the flow of H+ downwards this concentration slope through a fifth enzyme complex, known as ATP synthase, leads to a modify in the structure of the synthase, causing the synthesis and release of ATP.
In cells that are using energy, the turnover of ATP is very high, and then these cells contain high levels of ADP. They must therefore consume large quantities of oxygen continuously, and then as to have the energy necessary to phosphorylate ADP to form ATP. Consider, for example, that resting skeletal muscles employ about 30% of a resting adult's oxygen consumption, merely when the same muscles are working strenuously, they account for almost 90% of the total oxygen consumption of the organism.
Experiment has shown that ii.five–three ATP molecules are formed for every molecule of NADH oxidized in the electron transport chain, and i.v–ii ATP molecules are formed for every molecule of FADH2 oxidized. Table \(\PageIndex{one}\) summarizes the theoretical maximum yield of ATP produced by the complete oxidation of 1 mol of acetyl-CoA through the sequential activeness of the citric acid cycle, the electron ship chain, and oxidative phosphorylation.
| Reaction | Comments | Yield of ATP (moles) |
|---|---|---|
| Isocitrate → α-ketoglutarate + CO2 | produces one mol NADH | |
| α-ketoglutarate → succinyl-CoA + COii | produces 1 mol NADH | |
| Succinyl-CoA → succinate | produces ane mol GTP | +one |
| Succinate → fumarate | produces 1 mol FADHtwo | |
| Malate → oxaloacetate | produces 1 mol NADH | |
| ane FADH2 from the citric acid cycle | yields two mol ATP | +ii |
| 3 NADH from the citric acrid cycle | yields 3 mol ATP/NADH | +9 |
| Net yield of ATP: | +12 | |
Concept Review Exercises
- What is the principal office of the citric acid cycle?
- Two carbon atoms are fed into the citric acrid wheel every bit acetyl-CoA. In what grade are two carbon atoms removed from the wheel?
- What are mitochondria and what is their function in the jail cell?
Answers
- the consummate oxidation of carbon atoms to carbon dioxide and the formation of a high-energy phosphate chemical compound, free energy rich reduced coenzymes (NADH and FADH2), and metabolic intermediates for the synthesis of other compounds
- as carbon dioxide
- Mitochondria are small organelles with a double membrane that contain the enzymes and other molecules needed for the production of about of the ATP needed by the body.
Key Takeaways
- The acetyl group of acetyl-CoA enters the citric acid cycle. For each acetyl-CoA that enters the citric acrid wheel, 2 molecules of carbon dioxide, 3 molecules of NADH, 1 molecule of ATP, and one molecule of FADHtwo are produced.
- The reduced coenzymes (NADH and FADH2) produced by the citric acid cycle are reoxidized by the reactions of the electron transport chain. This series of reactions besides produces a pH gradient beyond the inner mitochondrial membrane.
- The pH gradient produced by the electron transport chain drives the synthesis of ATP from ADP. For each NADH reoxidized, 2.5–3 molecules of ATP are produced; for each FADH2 reoxidized, 1.5–2 molecules of ATP are produced.
Exercises
-
Replace each question marking with the correct compound.
- \(\mathrm{?\xrightarrow{aconitase}isocitrate}\)
- \(\mathrm{?\, +\, ? \xrightarrow{citrate\: synthase} citrate + coenzyme\: A}\)
- \(\mathrm{fumarate \xrightarrow{fumarase}\, ?}\)
- \(\mathrm{isocitrate + NAD^+ \xrightarrow{?} \alpha\textrm{-ketoglurate} + NADH + CO_2}\)
-
Supercede each question mark with the correct chemical compound.
- \(\mathrm{malate + NAD^+ \xrightarrow{?} oxaloacetate + NADH}\)
- \(\mathrm{?\, +\, ? \xrightarrow{nucleoside\: diphosphokinase} GDP + ATP}\)
- \(\mathrm{\textrm{succinyl-CoA} \xrightarrow{\textrm{succinyl-CoA synthetase}} \,?\, +\, ?}\)
- \(\mathrm{succinate + FAD \xrightarrow{succinate\: dehydrogenase}\, ? + FADH_2}\)
-
From the reactions in Exercises 1 and 2, select the equation(s) by number and letter of the alphabet in which each type of reaction occurs.
- isomerization
- hydration
- synthesis
-
From the reactions in Exercises 1 and 2, select the equation(s) by number and alphabetic character in which each blazon of reaction occurs.
- oxidation
- decarboxylation
- phosphorylation
-
What like role do coenzyme Q and cytochrome c serve in the electron transport chain?
-
What is the electron acceptor at the end of the electron send concatenation? To what product is this compound reduced?
-
What is the function of the cytochromes in the electron transport chain?
-
- What is meant by this statement? "Electron send is tightly coupled to oxidative phosphorylation."
- How are electron send and oxidative phosphorylation coupled or linked?
Answers
-
- citrate
- oxaloacetate + acetyl-CoA
- malate
- α-ketoglutarate hydrogenase complex
-
- reaction in 1a
- reaction in 1c
- reaction in 1b
-
Both molecules serve as electron shuttles between the complexes of the electron send chain.
-
Cytochromes are proteins in the electron transport chain and serve as one-electron carriers.
Source: https://chem.libretexts.org/Courses/Saint_Marys_College_Notre_Dame_IN/CHEM_118_(Under_Construction)/CHEM_118_Textbook/12%3A_Metabolism_(Biological_Energy)/12.4%3A_The_Citric_Acid_Cycle_and_Electron_Transport
Posted by: brooksimalk1993.blogspot.com

0 Response to "Based On The Animation, How Many Electron Carriers Are Reduced In The Krebs Cycle Only?"
Post a Comment